El negocio de los bebés
Para las relativamente pocas parejas que experimentan infertilidad, la fertilización in vitro es el único medio para concebir un bebé. Entonces, ¿por qué tantos otros también utilizan las tecnologías de reproducción asistida? Dos libros excelentes tratan sobre el impacto de la FIV y el cribado embrionario en el futuro de la familia y de la sociedad.
The Pursuit of Parenthood: Reproductive Technology from Test-Tube Babies to Uterus Transplants
Margaret Marsh y Wanda Ronner. 2019. Johns Hopkins University Press, Baltimore. 274 pages.
Fables and Futures: Biotechnology, Disability and the Stories We Tell Ourselves
George Estreich. 2019. MIT Press, Cambridge, MA. 219 pages.
Querer tener un bebé es algo natural, pero a veces eso de «fructificad y multiplicaos» no sucede tan naturalmente. La fertilización in vitro (FIV) le da una ayudita a la naturaleza al unir óvulos y espermatozoides fuera del cuerpo. El proceso se está volviendo tan común que descifrar el acrónimo FIV es casi tan innecesario como descifrar «NASA».
Desde el nacimiento de Louise Brown —primera bebé concebida por FIV— ocurrido en 1978 hasta ahora, llevamos ya casi nueve millones de criaturas concebidas de este modo. En todo el mundo, cada año se llevan a cabo más de dos millones de ciclos de FIV, los cuales no son simples ni baratos.
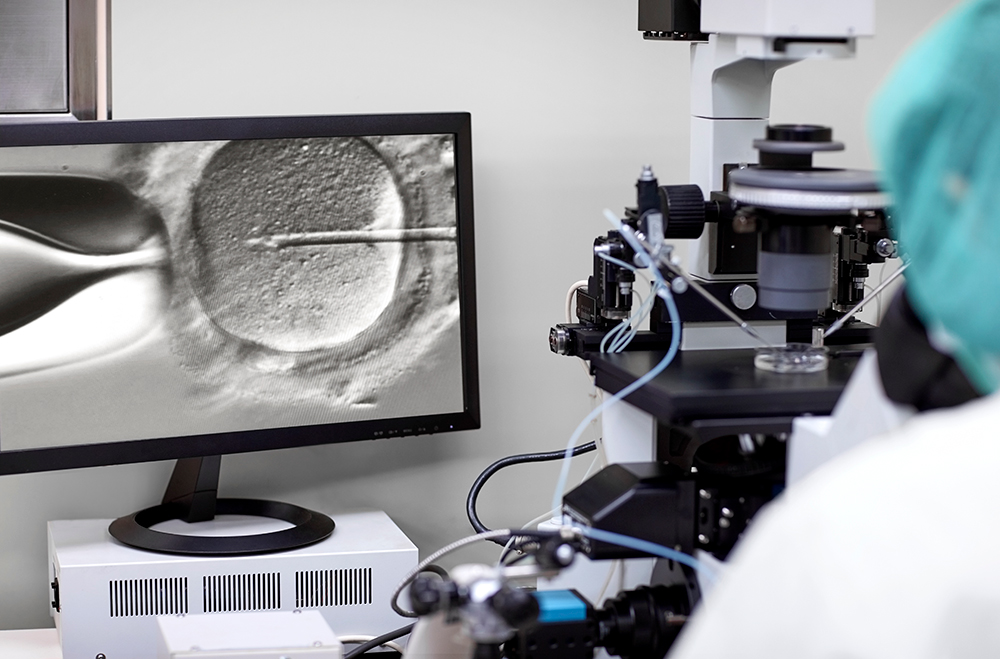
El procedimiento habitual para generar bebés suele iniciarse así: una mujer recibe inyecciones hormonales para estimular la ovulación de múltiples óvulos. Los óvulos maduros se extraen de los ovarios por medio de una aguja conectada a un dispositivo de succión. En un laboratorio, los embriólogos observan la condición de los óvulos recogidos y aparean los mejores con espermatozoides cuidadosamente examinados. Puede que congelen el esperma extra y los óvulos no fertilizados para usarlos posteriormente. Los óvulos fertilizados se dividen y crecen en una incubadora pequeña durante varios días.
Luego, en muchos casos, los embriones pasan por una evaluación genética. Las pruebas genéticas previas a la implantación (PGT, por sus siglas en inglés) conllevan examinar el genoma/ADN de una célula extraída del embrión en desarrollo para determinar anormalidades cromosómicas, enfermedades monogénicas y otros rasgos. Para los padres que son portadores de enfermedades genéticas, las PGT son una bendición, al detectar los embriones afectados antes de la implantación. Pero lo que se detecta es un blanco móvil. El síndrome de Down, el género, y de hecho cualquier rasgo vinculado a una secuencia de ADN se podría utilizar como un indicador para decidir si un embrión es adecuado para la implantación. Y por supuesto, a medida que la edición de la línea germinal y la manipulación de los rasgos se expandan en el futuro, esos embriones serán necesariamente evaluados para garantizar que las modificaciones previstas tengan éxito.
Por último, uno o más de los embriones se introducen en el útero de la mujer que es la fuente biológica del óvulo o de una madre sustituta gestacional. (Los padres potenciales pueden también usar los óvulos y/o el esperma de donantes en lugar de los propios). Actualmente, el índice general de éxito se calcula en 30% o menos. Pero si todo va bien, un embrión se implantará de por sí en la pared uterina y, nueve meses después, esta secuencia al estilo de Rube Goldberg resultará en el nacimiento de un nuevo miembro en la familia.
Al igual que los espermatozoides y óvulos sobrantes, todo embrión restante se puede congelar como una especie de reserva en caso de que no se produzca el embarazo. Determinar la cantidad exacta es imposible, pero con más de doscientos ochenta mil ciclos de FIV efectuados en los Estados Unidos en solo 2017 (según las cifras más recientes de los Centros Estadounidenses para el Control de las Enfermedades), algunos calculan que, actualmente, un millón o más de embriones sobrantes se están conservando criogénicamente en todo el país. Tras un embarazo exitoso, la suerte de esos embriones es frágil —¿adopción?, ¿abandono?, ¿utilización en investigación?— y su conservación es costosa.
Anualmente, alrededor de quinientos mil bebés se suman a la población mundial utilizando lo que en términos generales se denomina tecnología de reproducción asistida (TRA). En el contexto de más de ciento treinta millones de bebés nacidos cada año, esta no es una cifra particularmente elevada. Pero hay una creciente preocupación de que la fecundación in vitro conducirá a un nuevo tipo de humanidad diseñada, una nueva clase de planificación familiar. En ciertas maneras ya lo ha hecho.
Aunque la FIV ha constituido un milagro para los que lidian con la infertilidad, va mucho más allá de eso. Como una ventana a la condición genética del embrión, proporciona a los padres una manera de decidir en contra de enfermedades debilitantes, mas también sobre rasgos como el género o el color de ojos de su bebé, y hasta sobre su intolerancia a la lactosa. Cuanto más aprendamos sobre el genoma humano, más crecerá esa lista. Es más, como plataforma para la intervención genética a través de la edición de genes, la FIV resulta una posible vía de mejora genética, al menos para aquellos que pueden costearla.
Además, la FIV permite que una mujer pulse el botón de pausa en su reloj biológico. Al combinarla con la extracción del óvulo y la congelación, puede posponer la maternidad y seguir una carrera profesional u otras metas que se pueden alcanzar más fácilmente mientras no se tienen hijos.
La tecnología de reproducción asistida del «Salvaje Oeste»
Mientras avanzamos hacia la independencia reproductiva y el control genético, tanto las personas con o sin pareja como los científicos e historiadores se formulan preguntas importantes: ¿Deberían la congelación y la FIV convertirse en la forma normal de controlar el tiempo de la concepción? ¿Cómo decidiremos qué embriones descartar como anormales? ¿Son intercambiables los términos enfermedad y discapacidad? ¿Cuál será el papel de la regulación gubernamental?
En The Pursuit of Parenthood: Reproductive Technology from Test-Tube Babies to Uterus Transplants, la historiadora social Margaret Marsh (de la Universidad Rutgers) y la ginecóloga Wanda Ronner (de la Facultad de Medicina Perelman, en la Universidad de Pennsylvania) señalan que debemos pensar detenidamente sobre las razones sociales para practicar la tecnología de reproducción asistida de manera más amplia. Según ellas, los Estados Unidos en particular constituyen un «Oeste Salvaje» en gran medida no regulado, e insisten en recalcar que «un mercado no regulado en la reproducción no sirve bien a este país».
«Puede que no tengamos una bola de cristal para prever exactamente qué forma adoptaría un buen sistema regulador en este país, pero si no comenzamos ahora, probablemente no seremos capaces de domar el salvaje oeste de la medicina reproductiva»…
El problema —explican ellas— radica en una historia política que ha puesto la medicina en manos del mercado. «Estamos ahora a cuatro décadas de la decisión de la administración de Jimmy Carter de ignorar en lugar de lidiar con las ramificaciones éticas y políticas de la creación de embriones humanos fuera del cuerpo de una mujer. Las tecnologías se han multiplicado. Así también lo han hecho las preguntas médicas y éticas sobre ellas».
Señalan, además, que «en este país, las comisiones éticas van y vienen. Las recomendaciones de expertos en bioética y otros para formular políticas nacionales caen en oídos sordos en el Congreso».
Marsh y Ronner observan que la renuencia del congreso a formular regulaciones sobre la FIV surgió de la política del aborto. Su legalización en la decisión de 1973 de Roe v. Wade hizo de la medicina reproductiva un tercer carril: intocable, demasiado cargado políticamente para abordarlo. Como resultado, se prohibió que los estudios en relación con embriones humanos recibieran subvenciones federales para la investigación; se promulgaron moratorias y nunca se levantaron. Mientras el resto del mundo creaba normas y reglamentos que permitían la evolución de la medicina de FIV bajo la supervisión del gobierno, en los Estados Unidos los investigadores recurrieron a la financiación privada, y la financiación privada ha llevado a la mercantilización de la FIV y la TRA.
Según Marsh y Ronner —divididos por argumentos sobre cuestiones como el aborto, la igualdad de género y las definiciones de familia—, los Estados Unidos «automáticamente han permitido que el mercado determine la evolución de los servicios de reproducción asistida y el acceso a ellos».
Paciente y consumidor
Dado que en los Estados Unidos las clínicas de fertilidad con fines de lucro conducen y venden servicios de TRA, están siempre en busca de clientes nuevos y repetidos. Sea tocando el tambor del reloj biológico, presionando sobre la necesidad de evitar las enfermedades genéticas, o apelando al poder de la mujer de ejercer su independencia reproductiva, las clínicas comercializan para todas las mujeres considerándolas consumidoras potenciales de sus productos. Según Marsh y Ronner explican, «la medicina reproductiva del siglo XXI ha prosperado en el sector privado… porque por lo general las pacientes pagan de su bolsillo por la FIV y otras tecnologías avanzadas, y los centros de FIV pueden ser altamente lucrativos». Los investigadores de mercado predicen que el mercado mundial para la FIV en 2024 será de cuarenta mil millones de dólares; un aumento de diecinueve mil millones de dólares desde 2018.
En consonancia con las respectivas especialidades de sus autoras, The Pursuit of Parenthood sigue la evolución histórica de la TRA centrándose en los problemas de vida y de salud de la mujer. Se trata tanto de equidad social como del procurar formar una familia, y muestra que, desafortunadamente, no somos muy buenos ni para lo uno ni para lo otro. Según recalcan, la solución comienza con la atención médica reproductiva que no está sujeta al capricho del gobierno ni a merced del mercantilismo. «Ambas creemos que así como la atención médica es un derecho humano, la atención de la fertilidad se debería considerar parte de la atención médica básica de la mujer».
Marsh y Ronner dejan al lector preguntándose si hemos creado una solución tecnológica para una disfunción social (y engañosa al respecto). La congelación del óvulo, por ejemplo, se ha vendido a las mujeres jóvenes no solo como una manera de preservar los óvulos antes del tratamiento para enfermedades como el cáncer, sino también simplemente para posponer formar una familia. Según estas autoras, el mensaje ha sido que «los años fértiles de la mujer se les estaban yendo».
Y esto ha funcionado. Como recientemente la periodista de treinta y tantos años Natalie Lampert señalaba, con entusiasmo, en un artículo para Think de la NBC, la congelación de los óvulos, la FIV, la planificación de la carrera y el estilo de vida van naturalmente de la mano. «Para cierto grupo de mujeres —decía ella— la congelación de óvulos ha probado ser una herramienta empoderadora, transformando la vida personal de las mujeres de manera profunda al ofrecer una sensación de independencia aquí y ahora, a la vez que paz mental en términos de familia en el futuro».
Obviamente, ese «cierto grupo de mujeres» se refiere a mujeres jóvenes y adineradas. Sin embargo, al menos uno de ginecólogos insiste en que la congelación de óvulos «debería estar en el radar de toda mujer de entre veinte y treinta años», lo cual considera «una póliza de seguro para su salud reproductiva». Pero será una «póliza» cara, que actualmente oscila entre $6.000 y $20.000. Con un ciclo básico de FIV que cuesta de diez mil a veinte mil dólares adicionales (el cribado genético y/o la edición de genes podrían agregar miles más a la cuenta), la pregunta es quién podrá realmente participar en este nuevo mundo de planificación familiar. Aun para quienes pueden costearlo, un embarazo exitoso está lejos de ser algo seguro.
Esta realidad, a pesar del optimismo implícito de los vendedores de fertilidad, es una de las advertencias más fuertes de The Pursuit of Parenthood’s. Según las mencionadas autoras, la moraleja tácita de estas historias es «Tras gastar decenas de miles de dólares para congelar sus óvulos, una puede fácilmente terminar pobre e infértil». Esto así porque «hasta a las mujeres jóvenes, la congelación de óvulos no ofrece promesas».
Ingeniería social
En líneas generales, The Pursuit of Parenthood argumenta que no se está tratando lo que habría que tratar. «No vemos ni una palabra sobre la reingeniería del matrimonio como una sociedad más igualitaria, o la reingeniería del lugar de trabajo y de la sociedad en general para proporcionar los tipos de apoyos estructurales que permitirían a las mujeres tener hijos antes y aun así tener carreras exitosas».
Las autoras aportan un enfoque importante a estos difíciles problemas. Aunque su libro aplaude a la ciencia que ha aumentado la elección reproductiva de la mujer y lamenta las barreras sociales que frenan el progreso femenino, no profundiza en algunos de los aspectos más novedosos del control genético; por ejemplo, la evolución de los usos del cribado embrionario y la edición en la línea germinal utilizando tecnologías como CRISPR, y preguntas cada vez más apremiantes sobre la selección de embriones.
Ciertamente, Marsh y Ronner no son irracionalmente hostiles contra la TRA. Ambas creen que «es rol del médico prevenir el sufrimiento» y que usar estas tecnologías para ese fin no es lo mismo que crear un bebé de diseño. También reconocen la tensión entre lo que se debería eliminar y lo que no. ¿Cómo definimos el sufrimiento? ¿Deberíamos eliminar un embrión ante la probabilidad de que él o los padres sufran de alguna manera?
«Los defensores de los derechos de los discapacitados han argumentado que el deseo de tener un hijo sin una enfermedad genética puede conducir a la devaluación de las vidas de quienes viven con una discapacidad. Esta preocupación debe tomarse en serio».
Definiendo discapacidad
«Tal como muchos de nosotros, me encuentro entre una manera vieja de hacer las cosas y una manera nueva», escribe George Estreich en Fables and Futures: Biotechnology, Disability and the Stories We Tell Ourselves. «Pero la especie, también, está entre una manera vieja de hacer las cosas y una manera nueva; entre el pasado de la reproducción humana normal y las formas asistidas digitalmente actualmente en marcha».
Estreich es instructor de la Escuela de Escritura, Literatura y Cine de la Universidad Estatal de Oregon, no un experto en ciencia, fertilidad o bioética. Pero su hija Laura nació con el síndrome de Down, de modo que aporta una perspectiva particular al examinar cómo utilizar mejor las tecnologías que podrían eliminar condiciones como esta. «Estoy profundamente a favor de la ciencia —escribe—; pero si nuestras nuevas biotecnologías están para ayudarnos, debemos verlas claramente, y debemos también reconocer que, en su forma actual, reflejan y amplifican nuestros conceptos erróneos sobre la discapacidad y nuestra devoción a la idea a menudo destructiva de “normal”».
En Fables and Futures, Estreich analiza en profundidad cuestiones de relaciones humanas que Marsh y Ronner necesariamente pasan por alto. Ellas reconocen la «desvalorización» de las personas discapacitadas, pero para Estreich, esto es un hecho. Muchos escritores hablan sobre la ciencia de las posibilidades genéticas y el potencial para eliminar enfermedades, pero la mayoría de ellos escriben desde la cabeza; Estreich escribe desde el corazón. Al fin y al cabo, eliminar la enfermedad genética habría significado eliminar a Laura.
Esta es una lectura obligada para cualquier persona interesada en las posibles consecuencias de la TRA. Aunque Estreich se centra en el síndrome de Down, sus argumentos se relacionan con la FIV y, en particular, con el cribado del embrión. ¿Qué si una pareja quiere varones en lugar de nenas? Todos los embriones femeninos se mantendrían congelados, dejados de lado para favorecer a los que llevan un cromosoma Y. Ese tipo de selección parental se permite porque la distinción entre discapacidad y deseabilidad es maleable y solo lo será más en esta nueva era de inspección genética. Fables and Futures nos ayuda a pensar en las opciones que tenemos ante nosotros y en las venideras.
¿Una persona o una proyección?
¿Qué vemos cuando notamos a alguien con una discapacidad? Estreich dice que a menudo se trata de lo que esperamos ver: una proyección de nuestro propio prejuicio, o juicio; incluso de nuestra imaginación. Inevitablemente, la gente miraría fijamente al «fantasma abrazable» que era su hija: «una forma vaga, un diagnóstico con personalidad, una mezcla de dulzura y tragedia, de ángeles y defectos cardíacos y edad materna».
«Esa es una manera de imaginar el síndrome de Down, y no la peor manera, pero esconde al individuo. La proyección, el fantasma, obscurece al niño».
Que albergamos prejuicios es una realidad difícil de entender y admitir, pero es una idea clave si hemos de hallar el camino que debemos seguir. «Este libro está escrito en un momento de provisionalidad —dice Estreich— y examina las historias que nos contamos, los guiones que traemos a lo sin guión, las fábulas que ayudan a crear nuestro futuro». Y aquí está el punto: Hablamos acerca de personas aun cuando hablamos acerca de embriones. «Dada la biotecnología que puede seleccionar y dar forma a lo que somos, tenemos que imaginar, tan ampliamente como sea posible, qué significa pertenecer».
Podemos saber que alguien con síndrome de Down es una persona, pero ahora estamos en condiciones de garantizar que no nazcan más personas así. ¿Deberíamos? Es un lugar extraño y desafiante para estar, pero aquí estamos. El examen prenatal «conduce la manera en que pensamos y hablamos sobre las personas con síndrome de Down: lejos de una discusión de ciudadanos con derechos y hacia una discusión de posibilidades, potencialidades y riesgos —según señala Estreich—, ellos ocupan una especie de limbo de valor humano. Se discute sobre ellos —a menudo erróneamente— en términos de su efecto en otros, más bien que en términos de sus oportunidades y esperanzas». Para él, es asunto de dignidad: «Para muchas personas con discapacidades, las suposiciones de que sus vidas son menos valiosas, o definidas por el sufrimiento, son asaltos a la dignidad tan palpables como cualquier hecho físico».
Declaración Universal de la UNESCO sobre el genoma humano y los derechos humanos
La Conferencia General,
[…]
Reconociendo que las investigaciones sobre el genoma humano y sus aplicaciones abren inmensas perspectivas de mejoramiento de la salud de los individuos y de toda la humanidad, pero destacando que deben al mismo tiempo respetar plenamente la dignidad, la libertad y los derechos de la persona humana, así como la prohibición de toda forma de discriminación fundada en las características genéticas,
Proclama los principios siguientes y aprueba la presente Declaración.
A. La dignidad humana y el genoma humano
Artículo 1
El genoma humano es la base de la unidad fundamental de todos los miembros de la familia humana y del reconocimiento de su dignidad y diversidad intrínsecas. En sentido simbólico, el genoma humano es el patrimonio de la humanidad.
Artículo 2
(a) Cada individuo tiene derecho al respeto de su dignidad y derechos, cualesquiera que sean sus características genéticas.
(b) Esta dignidad impone que no se reduzca a los individuos a sus características genéticas y que se respete su carácter único y su diversidad.
Artículo 3
El genoma humano, por naturaleza evolutivo, está sometido a mutaciones. Entraña posibilidades que se expresan de distintos modos en función del entorno natural y social de cada persona, que comprende su estado de salud individual, sus condiciones de vida, su alimentación y su educación.
Artículo 4
El genoma humano en su estado natural no puede dar lugar a beneficios pecuniarios.
El punto de Estreich no se limita al síndrome de Down. Gracias a la FIV y las TPG (tecnologías postgenómicas) se puede sustituir cualquier rasgo. En el contexto de la biotecnología, las personas discapacitadas se ven como «resultados a evitar», concluye. «Sus identidades son ocluidas por diagnósticos o estereotipos, su interioridad no se reconoce ni se destaca, sus emociones se simplifican por diseño, y rara vez se las consulta». Sin embargo, paradójicamente —sigue explicando Estreich— ellas son esenciales. Como dispositivo retórico, la discapacidad ofrece la base para el desarrollo y uso de la tecnología, es decir, una promesa central de la tecnología es que la discapacidad será reparada o prevenida».
Los próximos 40 años
El experto en bioética Leon Kass predijo, a principios de esta era de la FIV, que el milagro de la vida daría paso a nuestra imaginación y a nuestro deseo de juguetear. «Con la fertilización in vitro —dijo— el embrión humano emerge por primera vez de la oscuridad natural y la privacidad del vientre de su propia madre, donde se esconde en el misterio, a la luz brillante y la publicidad absoluta del laboratorio del científico, donde será tratado con racionalidad inquebrantable, ante el ojo inteligente y desvergonzado de la mente y bajo el obediente e igualmente inteligente toque de la mano».
¿Cómo decidiremos seguir adelante? «Nuestro modo de tratar a la gente depende de lo que pensamos de ella; y lo que pensamos es, a su vez, revelado e influenciado por lo que decimos; razón por la cual la retórica asociada a la biotecnología —la retórica de los creadores—importa», señala Estreich.
«Como escritor, pienso acerca del futuro. Como padre pienso acerca del futuro de Laura. Últimamente, ese futuro parece más frágil».
El tiempo parece moverse más rápido a medida que las tecnologías de reproducción asistida toman un control cada vez más invasivo del embrión. A pesar de las distracciones de este mundo agitado, hay decisiones que tomar. ¿Demasiado complicado? Quizás se las dejemos a las máquinas. Como plantea Estreich: «Quizás, dados los retos conceptuales de analizar las diferencias entre enfermedad y discapacidad y variación, dejaremos que los algoritmos hagan el trabajo por nosotros, viendo como “anormal” lo que podemos detectar o afirmar que detectamos».
Esa sería la opción más aterradora. Como con todas las tecnologías, tenemos que considerar las consecuencias de una manera práctica. Estreich usa The Amazing Spider Man (2012) para resaltar este punto. La película gira en torno a los esfuerzos de un científico para modificar genéticamente el poder regenerativo de los reptiles en el genoma humano a fin de que las extremidades puedan volver a crecer. Suena bien; aunque, como es de esperar, el mejor de los planes de aprovechar la biología en beneficio humano acaba espantosamente mal: el empalme de genes no funciona. Por supuesto, esto es solo cosa de atroz ciencia ficción en libro de historietas; o no. La arrogancia simplemente aparece más descaradamente en las películas. Y ese es el punto de Estreich.
«La trama [de la película] refleja la ley de las consecuencias no deseadas —explica—, los peligros de mezclar ganancias e investigación, y… una decisión de principios de no dar seguimiento a un descubrimiento».
La lección más importante, según Estreich, es que si no prestamos atención a lo que está pasando en el mundo, somos, cada uno, cómplices del mal que abunda. «No se trata de solo un mal usuario, sino de miles de millones de consumidores comunes, siguiendo deseos comunes —conduciendo automóviles, deseando bebés sanos, yendo al cine—, dando forma a nuestro mundo».



